Because of their excellent selectivity and effectiveness, peptides are also widely acknowledged as being generally safe and well-tolerated. It follows that pharmaceutical research and development (R&D) is paying more attention to peptides, and over 140 potential treatments are now in clinical studies. Because the easy peptide targets have already been taken advantage of, it’s time to look for fresh approaches beyond what’s been done with standard peptide design. A few examples of these techniques include multifunctional peptides and peptide drug conjugates that penetrate cells. There is a lot of discussion on the present state of peptides as medicines, their strengths and drawbacks, and the new potential in peptide drug development that are opening up.
Introduction
Because of their inherent shortcomings, naturally occurring peptides are not always ideal for use as conventional treatments. These disadvantages include low chemical and physical stability as well as a short half-life in the blood. These issues need to be solved if they are to be used as medications for the general public. Therapeutic peptides with ‘conventional design,’ which we refer to as a solution to some of these problems, have had some success. The peptide industry is seeing a variety of new technologies emerge in addition to classic peptide design, which provides potential and future prospects. These include peptides with several functions and peptides that penetrate cells, as well as drug conjugates based on peptides and technologies aimed at finding new ways to administer drugs. This paper presents an argument that peptides will have significant promise as medicines in the future.
Peptide-based conventional methods
In the evolution of signal transduction molecules, peptides for sale have shown to be very potent, causing significant physiological consequences. For the most part, these drugs have poor physical and chemical qualities that make them unsuitable for use as medications. In order to overcome these shortcomings, classic rational peptide therapies design has centered on strategies that address them.
Peptide therapies designed rationally
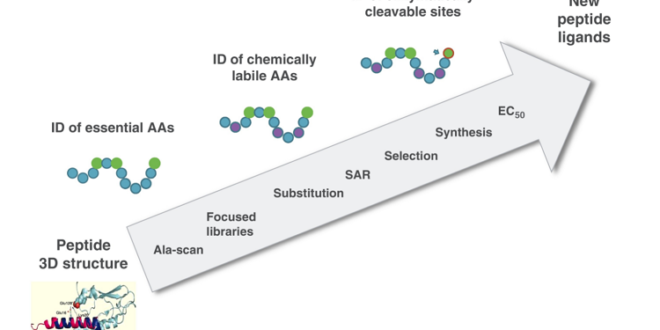
The secondary and tertiary structure of a peptide may be derived from its crystal structure via rational design. The structure-activity relation (SAR) is then constructed in consecutive phases to identify important amino acids as well as probable replacement sites using input from different studies, such as alanine substitutions (Ala-scan) and tiny targeted libraries. A crucial stage in this procedure is to identify chemically labile amino acids that are susceptible to isomerization, glycosylation, or oxidation, all of which should be avoided. This is particularly true when the ultimate result is a liquid medication formulation.
Additionally, rational peptide medication design involves enhancing the physicochemical features of endogenous peptides, which have a propensity to agglomerate and are not always water-soluble. Using substitutions or N-methylation of certain amino acids as chemical design solutions to minimize aggregation includes the corruption of hydrophobic regions. If a particular peptide therapeutic candidate has solubility concerns, the emphasis is usually on the charge distribution and the isoelectric point (pI) of the peptide in relation to the pH of the final formulation sought.
Because of the inherent physicochemical hurdles posed by peptides as medications, inferior solutions have been found for patients and clinicians in certain circumstances. Treatment of severe hypoglycemia by the use of glucagon is one example of this. Today, the sole glucagon rescue kit for the treatment of unconscious patients with hypoglycemia is sold, and it comes with an unopened vial of carefully reconstituting the peptide powder 30 times with mild agitation before it is injected subcutaneously to the unresponsive patient.
 HammBurg Be informed with latest news, reviews, entertainment, lifestyle tips, and much more.
HammBurg Be informed with latest news, reviews, entertainment, lifestyle tips, and much more.